Effective Training of deep neck flexors: Manual therapy to "switch on" the muscles!
CLINIAL SCENARIO
Neck pain is a common musculoskeletal disorder with a global prevalence of around five percent (women 5.8%, men 4.0%) [1]. It is a disabling condition with one of the highest socioeconomic burdens globally and is forecast to escalate with the ageing population [2].
Motor control of the deep and superficial neck flexors are usually reduced in chronic neck pain, with a delayed onset of neck muscle contraction during upper limb movements. Furthermore, there is an altered muscle activation pattern, showed by decreased deep neck flexors and an increased activity of the superficial cervical flexors during low load tasks [3], [4].
In MRI investigations, significant smaller Mm. longus colli, longus capitis, sternocleidomastoideus (SCM), levator scapulae, semispinalis capitis, trapezius were found in patients suffering from non-specific neck pain (NSNP) compared to healthy controls. In patients with WAD fatty infiltration was revealed in Mm. SCM, trapezius, semispinalis and splenius capitis, obliquus capitis inferior compared to patients with NSNP or healthy controls [5].
Clinical practice guidelines for neck pain recommend testing active and passive mobility, deep neck flexors, cervico-scapulo-thoracic muscles and motor control [6].
As muscles are often impaired in neck conditions, their recovery is crucial. There is no consensus about which exercise is the most effective [7], [8], currently, the combination of exercise and manual therapy seems to be beneficial [6], [9]–[11]. The question arises, if manual mobilization or manipulation enhances the efficiency of muscle training? Sterling et al. 2001 already showed positive results manual therapy concerning pressure pain threshold, sympathetic activity and activity pattern of the deep neck flexors [12].
FOCUSSED CLINICAL QUESTION
Does spinal mobilization or manipulation have an impact on the activity of the deep neck flexors?
SEARCH STRATEGY
In April 2023 the following search literature strategy was used to find suitable papers (Fig. 1):
|
Terms used to guide the search strategy |
|
· Patient Group: patients with neck conditions/asymptomatic individuals with forward head posture · Intervention: cervical mobilization or cervical manipulation or spinal mobilization or spinal manipulation · Comparison: before vs. after intervention · Outcome(s): activity deep neck flexors, strength deep neck flexors, endurance deep neck flexors |
Databases:
- PubMed – National Library of Medicine
- Cochrane Library
- PEDro – Physiotherapy Evidence Database
|
Inclusion Criteria |
|
· Measurement of the deep cervical flexors (activity, strength, endurance) · Humans · Asymptomatic subjects · Neck conditions (forward head posture, neck pain, cervical radiculopathy) · Cervical mobilization or cervical manipulation, spinal mobilization or spinal manipulation · Papers in English or German language |
|
Exclusion Criteria |
|
· Missing measurement of the deep cervical flexors or unclear measurement (e.g., cranio–cervical flexion angle) · Serious pathologies such as cancer · Other treatment techniques than spinal mobilization or manipulation, such as neurodynamic mobilization or trigger point therapy · Publication no older than 12 years · Case studies, case series |
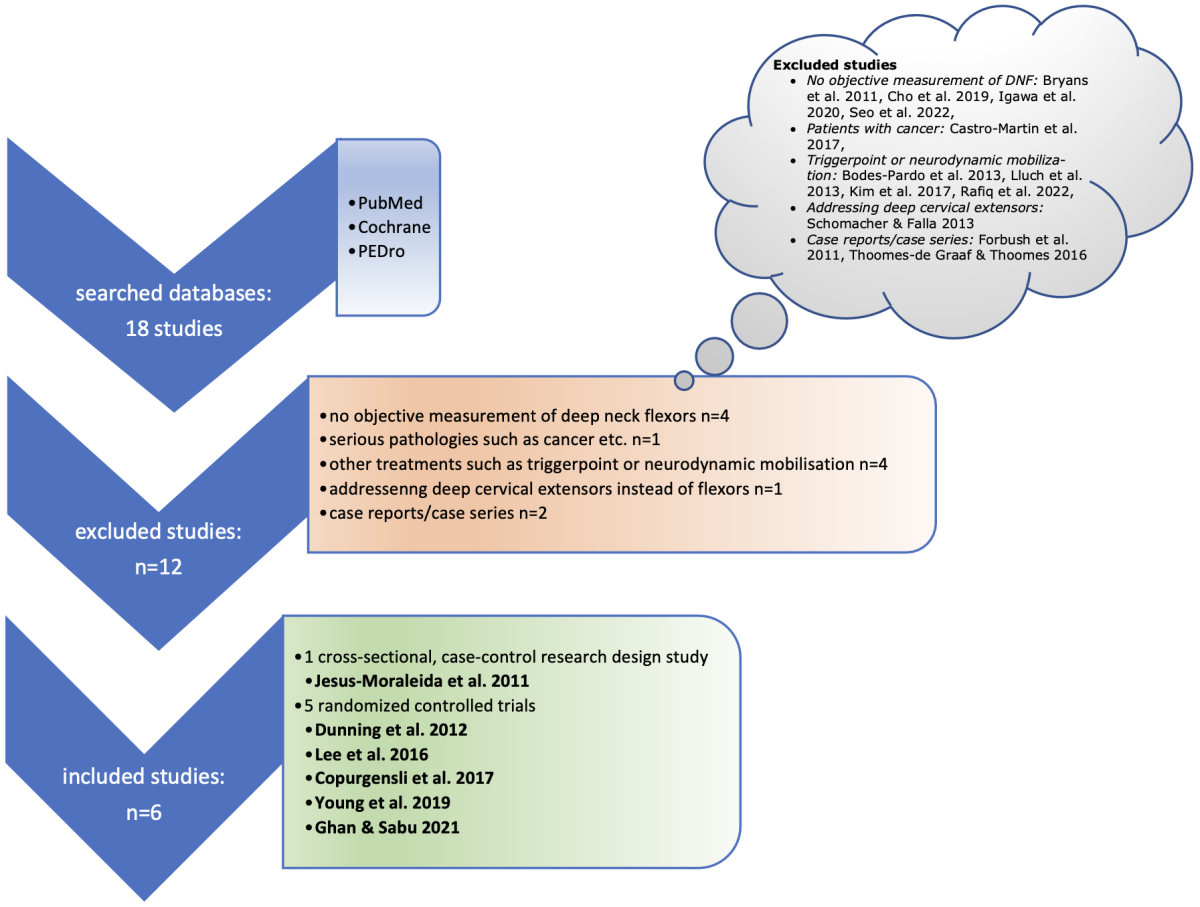
Figure 1: Search strategy
RESULTS OF THE STUDIES
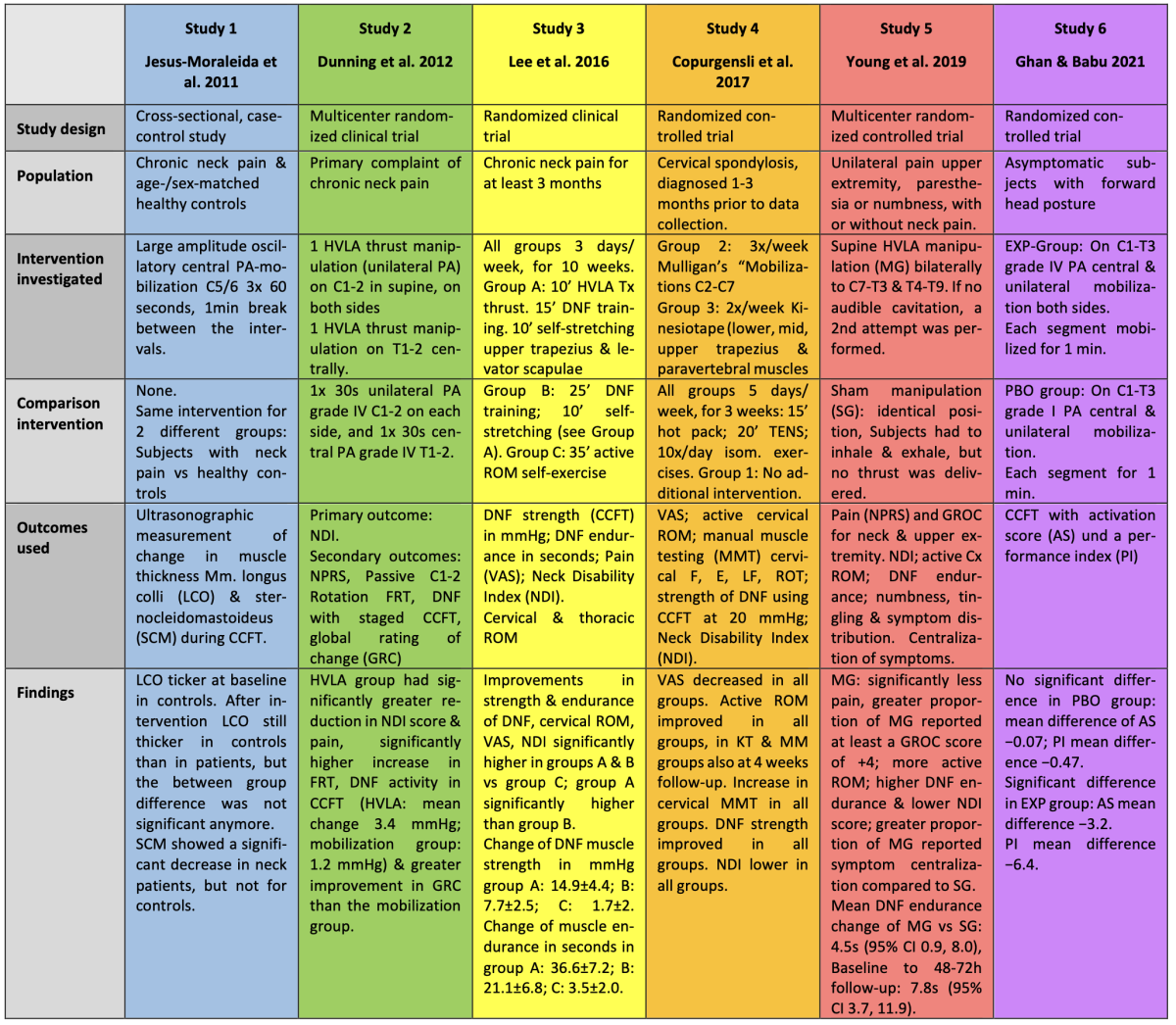
Table 1: Characteristics and results of included studies
DISCUSSION
The aim was to examine the effect of spinal manipulation on the activity of deep neck flexors. All included studies had as a primary or secondary outcome an objective measurement of deep neck flexor activity. A limitation can be the heterogeneity of participants (asymptomatic, neck pain, radiculopathy) and also the different ways of measuring DNF activity (ultrasonography, staged CCFT, strength, endurance, different scores). One could argue, that in clinical situations patients present with different clinical patterns, from minor issues like mild neck pain, up to severe painful radiculopathy. Therefore, the heterogeneity of neck conditions is like a mirror of clinical practice. Every patient may show different issues in the physical examination, from this point of view, it is useful to have several ways to test the DNF muscles such as strength or endurance. Furthermore, it may be of use to know that different functions of the DNF muscles can be addressed with spinal manipulation or mobilization as an addition to appropriately selected active exercises according to the findings of the physical examination.
Jesus-Moraleida et al. 2011 [13] revealed an increase in thickness of M. longus colli (LCO) in healthy controls and subjects with neck pain, and a decrease in thickness of sternocleidomastoideus (SCM) in neck patients only, in ultrasonography during Craniocervical Flexion Test (CCFT) after PA-mobilization to the spinous process C5/6.
Mobilization seems to normalize activity patterns of cervical flexor muscles in neck pain patients and to increase motor activity of LCO. Long-term effects are unknown. The study contributes to the hypothesis that cervical mobilization has a short-term impact on the activity pattern of DNF. The results have to be interpreted with caution, as the authors already mentioned, there have been included rather young subjects with a mean age of below 30 years. The result might not be the same in an older population – as elderly people show a different muscle activation pattern during CCFT, using higher levels of SCM than young adults [14]. Another detail has to be discussed: In a clinical setting, therapists usually choose the most painful or stiff segment, however in this study in every subject only the segment C5/6 was mobilized. It would have been interesting, if mobilizing the most obvious segment would have led to a higher difference in muscle thickness. Although the study was published already 12 years ago, it gives an interesting insight into visible changes of the neck flexors after mobilization.
Dunning et al. 2012 [15] compared high velocity low amplitude (HVLA) thrust manipulation unilateral C1/2 on both sides and centrally on T1/2 with non-thrust mobilization (30s PA mobilization, grade IV [16] on the same segments) in patients with mechanical neck pain; 48h after the intervention, the HVLA group showed significantly higher improvement in the CCFT.
Although the authors explain, that the short duration time of 30s in the mobilization group was to avoid longer contact compared to HVLA group, it seems not applicable to a clinical situation. Mobilizations are usually applied for a few minutes or in several sets of 30s-2min. However, if already such a short intervention leads to a minor increase in DNF motor activity (1.2 mmHg [95% CI: 0.64, 1.8]), longer application in future studies could be promising. Suggesting further studies to examine the effectiveness of different types and dosages of manual therapy and including long-term follow-up data collection was already mentioned by the authors. A third group with contact only could be a useful control group for non-thrust mobilization. As already mentioned in the discussion of the study of Jesus-Moraleida et al. 2011 [13], manipulation or mobilizing always the same segments (C1-2 & T1-2) is not reflecting clinical situations where the most obvious segment is chosen. A question mark is, why is a cracking sound needed for a successful manipulation? Relying on a cracking sound to decide if a manipulation is effective, is not supported by research findings – effects are not dependent from cracking sounds [17]–[19].
In patients with chronic neck pain, Lee and Kim 2016 [20] compared thoracic HVLA manipulation (group A) on hypomobile segments, DNF training, Trapezius/Levator scapulae stretching, with a group B performing DNF training and Trapezius/Levator scapulae stretching alone and a group C of active ROM exercises only – all interventions were performed 3x per week over a 10 weeks period. The results of DNF muscle strength and endurance are significantly higher in the combined HVLA with DNF training than DNF training alone, and DNF training was significantly more successful than active ROM exercise.
The authors already mentioned the rather small sample size as a limitation, they suggest further studies investigating the long-term effect with a larger number of participants. Another limitation is the missing demographic data about participants, only onset time was provided. The combination of manipulation and DNF training is what is usually done in clinical setting. However, from a clinical view 3 sessions per week over a period of 10 weeks seems quite a high number of treatments, in total the subjects had 30 sessions. This is not very cost-effective for the health system and not realistic in clinical setting. After a few treatments patients should be able to exercise at home/at work to regain self-efficacy [21]. Nevertheless, this study underlines the importance of an additional manipulation as it seems to have a relevant effect. Strength improved almost as double as in the “training only” group and endurance improved as well clearly, despite the training group had 10min more time for exercises during the sessions than the HVLA group. Training seems to become more efficient with additional manipulation. A very positive point in this study is, they selected the hypomobile segments, which is comparable to clinical work.
A similar concern about treatment intensity arises in the study of Copurgensli et al. 2017 [22]. Their goal was to investigate the contribution of NAGs / SNAGs and Kinesiotape to an extensive (5x/week, during 3 weeks), conventional rehabilitation program, containing hot pack, TENS and isometric theraband exercises. Both additional therapy methods did not provide any benefit to the conventional rehabilitation program. Despite the authors conclude, that both the mobilization as well as the Kinesiotape group, showed an increased gain of DNF muscle strength and ROM during the treatment period, the table of results can not confirm this finding. Increase of DNF strength is similar in all groups, the control group just starts with a lower DNF strength but has the same improvement as the other two groups.
5 treatments a week over a period of 3 weeks is only possible in in-patient rehabilitation centers, there the question arises, does a patient with cervical spondylosis really need to stay in a rehabilitation clinic? A second concern is the very long duration of a single session, 15min hot pack followed by 20min TENS exceeds already average time of a treatment session of approximately 30min. This is not realistic for most physiotherapists, except for those working in an in-patient rehabilitation center. Furthermore, using hot pack and TENS is not recommended for nonspecific neck pain [6], [23], the only reason to use this intervention would be as a control intervention. This is the only study, which could not make any contribution to enhanced muscle activity after mobilization
Young et al. 2019 [24] assessed the immediate and short-term effects of a single session of HVLA Manipulation to C7-T3 and T4-T9 compared to a sham manipulation; deep neck flexor endurance was significantly higher in the HVLA group compared to the sham manipulation group directly after intervention and at 48-72h follow-up.
Although the manipulation group achieved a significant mean change in DNF endurance directly after the intervention (4.5s) and even higher at 48-72h follow up (7.8s), the authors doubt if the change was clinically meaningful, as the minimal detectable change (MDC) measured in patients with neck pain would be 16.2s. However, even if it might be below the MDC, 4.5-7.8s are easily measurable with a stopwatch, and this effect can be used for a more efficient training of the DNF muscles. The authors mention, that the patient’s beliefs might have had an influence on the outcomes, as they recognized which kind of treatment they received. Manipulation of C7-T3 and T4-T9 was used, as the lower cervical spine functionally includes T1-4 as well [25], this can still be described as local manipulation. T4-9 seems to be far away from the cervical spine. However, also in other studies thoracic manipulations led to remote effects, for example in the shoulder muscles activity in patients with subacromial pain [26]. For immediate and short-term benefit, manipulation of the cervicothoracic junction and mid thoracic spine seems to be an effective early treatment option. Especially mid thoracic spine manipulation can be useful in clinical situations, to not provoke any symptoms of the upper extremity. Like Dunning et al., they rely on a cracking sound as decision that the manipulation was effective, as already mentioned, effects are not dependent from cracking sounds [17]–[19].
Ghan & Babu 2021 [27] presented an immediate effect of central and unilateral PA mobilizations C1-T3 grade IV on DNF motor activity in asymptomatic subjects with forward head posture, which also exceeded the minimal detectable change (MDC) for activation score and the performance index clearly. The placebo group received grade I mobilizations on the same segments.
As a conclusion central/unilateral PA mobilization grade IV can be used to achieve more efficient DNF training. The authors suggest to investigate the mid-/long-term effect of grade IV mobilization on DNF muscle activation in further studies with asymptomatic subjects. Unilateral and central mobilization of all segments C1—T3 is quite a lot and not very practicable and efficient in clinical situations. Clinically, therapists would choose the most obvious movement direction and segment and its adjacent segments for mobilization. It would be interesting in further research to choose the most obvious segment to examine if this has a higher impact than mobilizing all segments. Another question is, why did the authors choose PA mobilizations and not AP [16], [25]? From clinical experience, subjects with forward head posture are usually in a more extended position of the upper and mid cervical spine and more (hyper)mobile into PA direction, but hypomobile into AP direction. Therefore, it could be beneficial in these group of participants to do AP mobilizations.
Out of the six included studies, five papers show an immediate improvement of DNF motor activity, such as ultrasonographic visible thickness, strength or endurance following manipulation or mobilization of the cervical or thoracic spine. One study did not find any additional benefit using mobilization compared to a control group. All of these studies have apart from their strengths also some limitations, which have already been discussed earlier. On the whole, we can conclude, that spinal manipulation or mobilization is a useful treatment option to enhance DNF motor activity and provide a more efficient training. Manipulation seems to be more effective than mobilization. Manipulation/mobilization should not be applied as a stand-alone treatment, always in combination with active therapy and education [6].
IMPLICATIONS FOR PRACTICE AND FUTURE RESEARCH
Clinicians should be aware of the positive effects of manipulation and mobilization. If DNF muscles are impaired, they have to be trained in therapy, in home-exercises and self-management [21] program. Exercise seems to be a substantial prevention for recurrent neck pain episodes [28], [29]. As an addition, manipulation or mobilization can improve efficiency of DNF training. Combination of manual techniques and training of DNF seems to be more effective than training alone. If there is a choice between manipulation and mobilization, according to the current evidence manipulation seems to be preferable.
Why can manipulations/mobilizations lead to more efficient training? Muscle activity can be inhibited in patients with neck pain[3], [30] by dysfunction of joints, pain, effusion, trauma and fatigue [29], [31]. Similar to arthrogenic muscle inhibition which is described to occur due to pain, effusion or joint dysfunction. These limiting factors have to be addressed to decrease inhibition and provide adequate training [32], [33]. If dysfunction and pain can be diminished – which can be addressed quickly with manipulation or mobilization – muscles have a higher chance to be regain normal activity. It is like if the plug is not completely in the power socket, the light is flickering and the light will not be as bright as steady light – if the muscle is inhibited, it can’t be trained as powerful as if would be “switched on” first.
How could we explain the effect of manual techniques? On the left/theoretical side of the brick wall [16], [25], there might be pain inhibition due to systemic neurophysiological responses caused by a mechanical force such as manipulation or mobilization [34], [35]. Neurophysiological effects of spinal manipulation are still investigated, there are effects reported about effects of spinal manipulation have been central neuroplastic alterations, changes in motor neuron excitability, enhanced muscle strength, improved cortical drive, activation of the descending pain modulation circuit and central sensitization [36].
On the right/clinical side of the brick wall [16], [25] we try to find stiffness or pain – where we could apply mobilization or manipulation. Moreover, we might find weakness, decreased endurance or proprioception to what we could tailor a training program or we recognize fear of movement, in which case the patient will need more education.
Ideas for future research are already mentioned in the discussion. Focus could be set on more clinically relevant interventions, such as choosing the most painful or stiffest segment, instead of mobilizing every segment. Furthermore, examining more realistic rehabilitation programs – like less frequency over a period of time. Comparison between manipulation, mobilization and a control group would be interesting, to investigate the effect of mobilization too. What is still missing are long-term results. However, from my point of view, it would be relevant to examine, how long the effect on DNF motor activity lasts after a single intervention, as this information could give an idea about treatment frequency.
|
Key points
|
REFERENCES
|
[1] D. Hoy et al., “The global burden of neck pain: estimates from the Global Burden of Disease 2010 study,” Ann. Rheum. Dis., vol. 73, no. 7, pp. 1309–1315, Jul. 2014, doi: 10.1136/annrheumdis-2013-204431. [2] T. Vos et al., “Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013,” The Lancet, vol. 386, no. 9995, pp. 743–800, Aug. 2015, doi: 10.1016/S0140-6736(15)60692-4. [3] D. L. Falla, G. A. Jull, and P. W. Hodges, “Patients With Neck Pain Demonstrate Reduced Electromyographic Activity of the Deep Cervical Flexor Muscles During Performance of the Craniocervical Flexion Test:,” Spine, vol. 29, no. 19, pp. 2108–2114, Oct. 2004, doi: 10.1097/01.brs.0000141170.89317.0e. [4] D. Falla, “Unravelling the complexity of muscle impairment in chronic neck pain,” Man. Ther., vol. 9, no. 3, pp. 125–133, Aug. 2004, doi: 10.1016/j.math.2004.05.003. [5] E. Van Looveren, B. Cagnie, I. Coppieters, M. Meeus, and R. De Pauw, “Changes in Muscle Morphology in Female Chronic Neck Pain Patients Using Magnetic Resonance Imaging,” Spine, vol. 46, no. 10, pp. 638–648, May 2021, doi: 10.1097/BRS.0000000000003856. [6] P. R. Blanpied et al., “Neck Pain: Revision 2017: Clinical Practice Guidelines Linked to the International Classification of Functioning, Disability and Health From the Orthopaedic Section of the American Physical Therapy Association,” J. Orthop. Sports Phys. Ther., vol. 47, no. 7, pp. A1–A83, Jul. 2017, doi: 10.2519/jospt.2017.0302. [7] J. Price, A. Rushton, I. Tyros, V. Tyros, and N. R. Heneghan, “Effectiveness and optimal dosage of exercise training for chronic non-specific neck pain: A systematic review with a narrative synthesis,” PLOS ONE, vol. 15, no. 6, p. e0234511, Jun. 2020, doi: 10.1371/journal.pone.0234511. [8] R. M. de Zoete, N. R. Armfield, J. H. McAuley, K. Chen, and M. Sterling, “Comparative effectiveness of physical exercise interventions for chronic non-specific neck pain: a systematic review with network meta-analysis of 40 randomised controlled trials,” Br. J. Sports Med., vol. 55, no. 13, pp. 730–742, Jul. 2021, doi: 10.1136/bjsports-2020-102664. [9] C. Bernal-Utrera, J. J. Gonzalez-Gerez, E. Anarte-Lazo, and C. Rodriguez-Blanco, “Manual therapy versus therapeutic exercise in non-specific chronic neck pain: a randomized controlled trial,” Trials, vol. 21, no. 1, p. 682, Dec. 2020, doi: 10.1186/s13063-020-04610-w. [10] S. Chung and Y.-G. Jeong, “Effects of the craniocervical flexion and isometric neck exercise compared in patients with chronic neck pain: A randomized controlled trial,” Physiother. Theory Pract., vol. 34, no. 12, pp. 916–925, Dec. 2018, doi: 10.1080/09593985.2018.1430876. [11] M. Sterling, R. M. J. de Zoete, I. Coppieters, and S. F. Farrell, “Best Evidence Rehabilitation for Chronic Pain Part 4: Neck Pain,” J. Clin. Med., vol. 8, no. 8, p. 1219, Aug. 2019, doi: 10.3390/jcm8081219. [12] M. Sterling, G. Jull, and A. Wright, “Cervical mobilisation: concurrent effects on pain, sympathetic nervous system activity and motor activity,” Man. Ther., vol. 6, no. 2, pp. 72–81, May 2001, doi: 10.1054/math.2000.0378. [13] F. R. Jesus-Moraleida, P. H. Ferreira, L. S. M. Pereira, C. M. Vasconcelos, and M. L. Ferreira, “Ultrasonographic Analysis of the Neck Flexor Muscles in Patients with Chronic Neck Pain and Changes After Cervical Spine Mobilization,” J. Manipulative Physiol. Ther., vol. 34, no. 8, pp. 514–524, Oct. 2011, doi: 10.1016/j.jmpt.2011.08.006. [14] S. Uthaikhup and G. Jull, “Performance in the cranio-cervical flexion test is altered in elderly subjects,” Man. Ther., vol. 14, no. 5, pp. 475–479, Oct. 2009, doi: 10.1016/j.math.2008.12.003. [15] J. R. Dunning et al., “Upper Cervical and Upper Thoracic Thrust Manipulation Versus Nonthrust Mobilization in Patients With Mechanical Neck Pain: A Multicenter Randomized Clinical Trial,” J. Orthop. Sports Phys. Ther., vol. 42, no. 1, pp. 5–18, Jan. 2012, doi: 10.2519/jospt.2012.3894. [16] E. Hengeveld, K. Banks, and G. D. Maitland, Eds., Maitland’s vertebral manipulation: management of neuromusculoskeletal disorders - Volume one, Eighth edition. Edinburgh: Elsevier Ltd, 2014. [17] J. A. Cleland, T. W. Flynn, J. D. Childs, and S. Eberhart, “The Audible Pop from Thoracic Spine Thrust Manipulation and Its Relation to Short-Term Outcomes in Patients with Neck Pain,” J. Man. Manip. Ther., vol. 15, no. 3, pp. 143–154, Jul. 2007, doi: 10.1179/106698107790819828. [18] D. W. Evans and N. Lucas, “What is ‘manipulation’? A reappraisal,” Man. Ther., vol. 15, no. 3, pp. 286–291, Jun. 2010, doi: 10.1016/j.math.2009.12.009. [19] J. Dunning, F. Mourad, M. Barbero, D. Leoni, C. Cescon, and R. Butts, “Bilateral and multiple cavitation sounds during upper cervical thrust manipulation,” BMC Musculoskelet. Disord., vol. 14, no. 1, p. 24, Dec. 2013, doi: 10.1186/1471-2474-14-24. [20] K.-W. Lee and W.-H. Kim, “Effect of thoracic manipulation and deep craniocervical flexor training on pain, mobility, strength, and disability of the neck of patients with chronic nonspecific neck pain: a randomized clinical trial,” J. Phys. Ther. Sci., vol. 28, no. 1, pp. 175–180, 2016, doi: 10.1589/jpts.28.175. [21] N. Hutting, V. Johnston, J. B. Staal, and Y. F. Heerkens, “Promoting the Use of Self-management Strategies for People With Persistent Musculoskeletal Disorders: The Role of Physical Therapists,” J. Orthop. Sports Phys. Ther., vol. 49, no. 4, pp. 212–215, Apr. 2019, doi: 10.2519/jospt.2019.0605. [22] C. Copurgensli, G. Gur, and V. B. Tunay, “A comparison of the effects of Mulligan’s mobilization and Kinesio taping on pain, range of motion, muscle strength, and neck disability in patients with Cervical Spondylosis: A randomized controlled study,” J. Back Musculoskelet. Rehabil., vol. 30, no. 1, pp. 51–62, Dec. 2016, doi: 10.3233/BMR-160713. [23] P. Kharel, J. R. Zadro, and C. G. Maher, “Physiotherapists can reduce overuse by Choosing Wisely,” J. Physiother., vol. 67, no. 3, pp. 151–155, Jul. 2021, doi: 10.1016/j.jphys.2021.06.006. [24] I. A. Young, F. Pozzi, J. Dunning, R. Linkonis, and L. A. Michener, “Immediate and Short-term Effects of Thoracic Spine Manipulation in Patients With Cervical Radiculopathy: A Randomized Controlled Trial,” J. Orthop. Sports Phys. Ther., vol. 49, no. 5, pp. 299–309, May 2019, doi: 10.2519/jospt.2019.8150. [25] G. D. Maitland, E. Hengeveld, K. Banks, and K. English, Maitland’s vertebral manipulation. Edinburgh; New York: Elsevier Butterworth-Heinemann, 2005. [26] A. K. Hegarty, M. Hsu, J.-S. Roy, J. R. Kardouni, J. J. Kutch, and L. A. Michener, “Evidence for increased neuromuscular drive following spinal manipulation in individuals with subacromial pain syndrome,” Clin. Biomech., vol. 90, p. 105485, Dec. 2021, doi: 10.1016/j.clinbiomech.2021.105485. [27] G. M. Ghan and V. S. Babu, “Immediate Effect of Cervico-thoracic Mobilization on Deep Neck Flexors Strength in Individuals with Forward Head Posture: A Randomized Controlled Trial,” J. Man. Manip. Ther., vol. 29, no. 3, pp. 147–157, May 2021, doi: 10.1080/10669817.2020.1834321. [28] T. F. De Campos, C. G. Maher, D. Steffens, J. T. Fuller, and M. J. Hancock, “Exercise programs may be effective in preventing a new episode of neck pain: a systematic review and meta-analysis,” J. Physiother., vol. 64, no. 3, pp. 159–165, Jul. 2018, doi: 10.1016/j.jphys.2018.05.003. [29] D. Falla, “Schlüsselprinzipien für das Training von Patienten mit Nackenschmerzen,” manuelletherapie, vol. 17, no. 01, pp. 7–13, Feb. 2013, doi: 10.1055/s-0033-1334131. [30] T. R. Stanton, H. B. Leake, K. J. Chalmers, and G. L. Moseley, “Evidence of Impaired Proprioception in Chronic, Idiopathic Neck Pain: Systematic Review and Meta-Analysis,” Phys. Ther., vol. 96, no. 6, pp. 876–887, Jun. 2016, doi: 10.2522/ptj.20150241. [31] U. Röijezon, N. C. Clark, and J. Treleaven, “Proprioception in musculoskeletal rehabilitation. Part 1: Basic science and principles of assessment and clinical interventions,” Man. Ther., vol. 20, no. 3, pp. 368–377, Jun. 2015, doi: 10.1016/j.math.2015.01.008. [32] J. T. Hopkins and C. D. Ingersoll, “Arthrogenic Muscle inhibition: A Limiting Factor in Joint Rehabilitation,” J. Sport Rehabil., vol. 9, no. 2, pp. 135–159, May 2000, doi: 10.1123/jsr.9.2.135. [33] G. Norte, J. Rush, and D. Sherman, “Arthrogenic Muscle Inhibition: Best Evidence, Mechanisms, and Theory for Treating the Unseen in Clinical Rehabilitation,” J. Sport Rehabil., pp. 1–19, 2021, doi: 10.1123/jsr.2021-0139. [34] J. E. Bialosky, M. D. Bishop, D. D. Price, M. E. Robinson, and S. Z. George, “The mechanisms of manual therapy in the treatment of musculoskeletal pain: A comprehensive model,” Man. Ther., vol. 14, no. 5, pp. 531–538, Oct. 2009, doi: 10.1016/j.math.2008.09.001. [35] J. E. Bialosky et al., “Unraveling the Mechanisms of Manual Therapy: Modeling an Approach,” J. Orthop. Sports Phys. Ther., vol. 48, no. 1, pp. 8–18, Jan. 2018, doi: 10.2519/jospt.2018.7476. [36] G. Gyer, J. Michael, J. Inklebarger, and J. S. Tedla, “Spinal manipulation therapy: Is it all about the brain? A current review of the neurophysiological effects of manipulation,” J. Integr. Med., vol. 17, no. 5, pp. 328–337, Sep. 2019, doi: 10.1016/j.joim.2019.05.004.
|
Comments
Thank you for this. Well done.
Cheers
Elly
- {{ error[0] }}